|
12/10/2023 0 Comments Caffeine structure with lone pairs 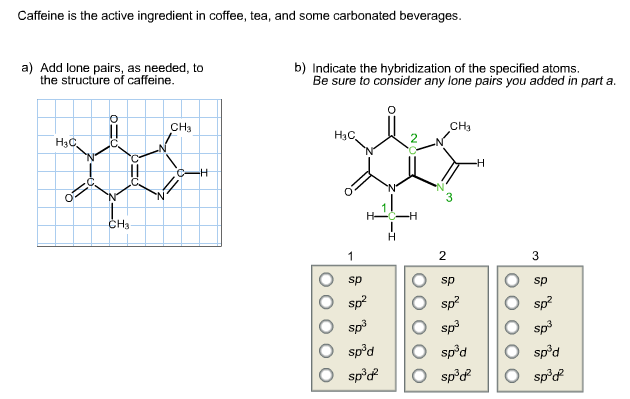
In particular, the first solvation shell water molecules of calcium are accessible to forming more » acceptor hydrogen bonds, and this results in an enhancement of a post-edge peak near 540 eV. For the divalent cations, we find that the hydrogen bonding characteristics of the more rigid magnesium first shell water molecules differ from those in the more flexible solvation shell surrounding calcium. Also observed are: (i) decrease in probability of salt contact ion pair as well as decrease in the solvent separated ion pair formation with increasing salt concentration, (ii) a modest second shell collapse in the water structure, and (iii) dehydration of hydrophobic atomic sites of caffeine on addition of =, have on the X-ray absorption spectrum of the respective solutions. The calculated hydrogen bond properties reveal that there is a modest decrease in the average number of water-caffeine hydrogen bonds on addition of NaCl salt. Science Chemistry The structure of caffeine, present in coffee and many soft drinks, is shown here. Furthermore, our calculated solvent accessible area values and cluster structure analyses suggest formation of higher order caffeine cluster on addition of salt. With increasing salt concentration, we observe enhancement of first peak height and appearance of a second peak in the caffeine-caffeine distribution function. The concentration of caffeine was taken almost at the solubility limit. Resonance shows the hypothetical images obtained when pi electrons or non-bonding electrons move from one orbital to another.The effect of salt concentration on association properties of caffeine molecule was investigated by employing molecular dynamics simulations in isothermal-isobaric ensemble of eight caffeine molecules in pure water and three different salt (NaCl) concentrations, at 300 K temperature and 1 atm pressure. So, sp 3 hybridization is placed on single-bonded carbon and sp 2 for double-bonded carbon.ĭ. In the skeletal structures of aspirin and caffeine, it is clear that only double bonds and single bonds are present. Hence, eight lone pairs (four on nitrogen and four lone pairs on oxygen) are present in the structure.Ĭ. Thus, the one lone pair is available on each nitrogen atom. In the case of caffeine, nitrogen has five valence electrons, and only three of them are involved in forming the bond. Hence, a total of eight lone pairs are present in the structure. structure by adding multiple bonds and lone pairs of electrons. is unlike the other two as it contains a lone pair of valence electrons. Thus, the two lone pairs are available on each oxygen atom. Identify the position(s) of multiple bonds in caffeine, and tell the hybridization of. But in any case, caffeine is NOT a very polar molecule (If it were it would not. In the case of aspirin, oxygen has six valence electrons, and only two of them are involved in forming the bond. In the skeletal structure of aspirin and caffeine, oxygen and nitrogen are heteroatoms. We recently launched MolPad, a commercial web-component for. Therefore, its molecular formula is C 8 H 10 N 4 O 2. The nitrogen atoms have a hybridization of sp3, due to the three covalent bonds and the lone pair that was added in part a. The skeletal structure of caffeine is formed by 8 C, 2 O, 4 N, and 10 H. The structure of caffeine is as follows: O //\\ NC-N-CC-N-C-N O The oxygen atoms have a hybridization of sp3, due to their four electron pairs (three covalent bonds and one lone pair). Therefore, its molecular formula is C 9 H 8 O 4. The skeletal structure of aspirin is formed by nine carbons, four oxygens, and eight hydrogens. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
